 Determination of estrogen receptor (ER), progesterone receptor (PgR), and HER2 status is mandatory for all patients with invasive breast cancer. Neoadjuvant application is preferred for response-guided systemic and local therapy, and if tumor size reduction is warranted for optimal surgical results or to assess the tumor’s response in vivo. Chemotherapy can be given before (neoadjuvant) or after (adjuvant) surgery with equivalent effects on outcomes, as first demonstrated by the NSABP-B18 trial. Systemic therapies for early breast cancer are highly effective the individual indication depends on patient characteristics, molecular subtype, tumor burden, and absolute risk of recurrence. Particularly, in view of the recent therapy advances, it will be critical to evaluate whether individualization of treatment concepts based on the biology of the non-pCR specimens is preferable to the initial therapy concept based on the pathology at primary diagnosis. Future research needs to address the impact of HR and HER2 status switch on therapy adaptation and on subsequent patient outcome. Yet, routine reevaluation of HR and HER2 status is not recommended in guidelines so far. These results are consistent with prior evidence. Conclusion: In a substantial number (27%) of non-pCR cases, a switch in biomarker status after completed neoadjuvant treatment was detected. Twenty cases had a switch in HR status, while 15 cases had a switch in HER2 status. In 35 cases (27%), a switch of either HR and/or HER2 status between the initial biopsy and the surgical specimen was detected. In 128 patients (52%), surgery revealed non-pCR after completed NAC. Results: 245 consecutive patients with primary invasive breast cancer, treated with NAC with/without targeted therapy between Januand December 31, 2019, at the LMU Breast Center, Munich, Germany, were identified. HR and HER2 status determined before NAC was compared with the corresponding receptor status determined in the surgical specimen after NAC. Pathological complete response (pCR) was defined as no invasive tumor in breast and lymph nodes (ypT0/is ypN0). 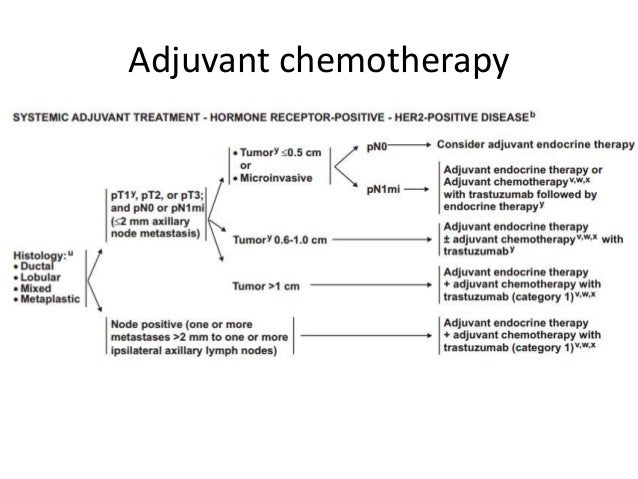
Methods: Tumor samples from patients without pathological complete response (non-pCR) were evaluated. Neoadjuvant TCH, non-anthracycline chemotherapy with single anti-HER2 regimen achieved high efficacy, with a good pCR rate and favorable tolerability in stage II or III HER2-positive breast cancer patients.Introduction: This project aimed to identify the frequency of a switch of hormone receptor (HR) and/or HER2 status after neoadjuvant chemotherapy (NAC) for early breast cancer. No symptomatic cardiac dysfunction occurred.Ĭonclusions. Treatment was generally well tolerated. Grade 3/4 neutropenia and anemia were uncommon (21.6% and 7.8%). 39.3%, p = 0.033). Three-year estimated DFS and OS rates were 81.3% and 93.0%, respectively. The pCR rate was 41.2% there was a significantly higher response in cT1-2 patients compared to cT3-4 ones (61.1% vs. The complete and partial clinical response of 51 patients were 33.3% and 58.8%, respectively. The secondary endpoints were DFS, OS, and toxicities. This retrospective study reviewed stage II–III HER2-positive breast cancer patients who received neoadjuvant docetaxel, carboplatin, and trastuzumab (TCH) at the Vietnamese National Cancer Hospital. The primary endpoint was the pCR rate which was defined as the absence of invasive tumor in the breast and axillary nodes (ypT0/is, ypN0). Purposes: To evaluate the efficacy and safety of neoadjuvant docetaxel, carboplatin, and trastuzumab (TCH) in the treatment of HER2-positive breast cancer in Vietnamese patients. The use of trastuzumab has been shown to improve pathological complete response (pCR), disease-free survival (DFS), and overall survival (OS). Neoadjuvant chemotherapy for HER2-positive breast cancer consists of a chemotherapy regimen plus trastuzumab with or without pertuzumab. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
